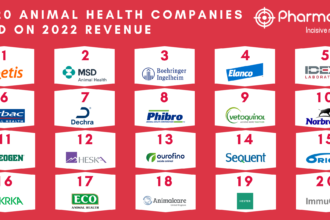
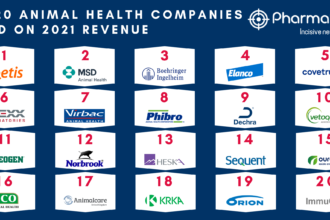
Quick Takeaways:
- Pegasus Laboratories (PRN Pharmacal) received full approval from the US FDA for KBroVet (potassium bromide chewable tablets) to control seizures linked to idiopathic epilepsy in dogs, marking a major milestone in veterinary neurology.
- KBroVet becomes the first fully FDA-approved drug specifically for canine idiopathic epilepsy and only the second animal health product to transition from conditional to full approval, reinforcing its clinical efficacy and safety profile.
- With features like once-daily dosing, liver-safe elimination, long half-life (~21 days), and chewable format, KBroVet offers a practical, compliance-friendly option for managing a condition affecting ~2.1M dogs in the US, improving outcomes for both pets and owners.
Why It Matters?
Full approval elevates KBroVet from a conditional option to the reference standard for potassium bromide in canine idiopathic epilepsy, giving vets greater confidence in its long‑term use.
Its once‑daily, chewable, liver‑safe profile makes it especially attractive for chronic management and combination therapy, which can drive wider adoption in general practice and neurology referral centers.
Source: PRnewswire